
Search
Selected Filter
Filter Results
Displaying 291–300 of 306 research results for “retinitis pigmentosa”
-

Jan 26, 2021
Genentech’s Faricimab Performs Well in Phase 3 Clinical Trials for Wet AMD
Eye On the Cure Research NewsCompany will seek marketing approval for the emerging therapy
-

Jan 7, 2021
REGENXBIO’s Wet AMD Gene Therapy Moving into Phase 3 Trials
Eye On the Cure Research NewsIn a Phase ½ clinical trial, the emerging treatment reduced the burden of regular anti-VEGF injections
-

May 28, 2020
Genentech’s Port Delivery System for Wet AMD Meets Primary Endpoint in Phase 3 Clinical Trial
Eye On the Cure Research NewsTiny implant provides continual delivery of anti-VEGF treatment for six months
-

May 15, 2020
Positive Interim Clinical Trial Results for Adverum’s Wet AMD Gene Therapy
Eye On the Cure Research NewsThe gene therapy is designed to reduce the treatment burden for patients
-

Dec 17, 2019
First Human Study in US for Induced Pluripotent Stem Cells to be Launched for Advanced Dry AMD
Eye On the Cure Research NewsCurrently there are no treatments for Advanced Dry AMD, also known as GA, which can lead to significant central vision loss
-

Jul 10, 2019
The Foundation’s Contributions to Genetic Research Published in Peer-Reviewed Journal
Eye On the Cure Research NewsThe nonprofit has emerged as a driving force in genetic research and treatment development for inherited retinal diseases.
-

Sep 18, 2018
Apellis Launches Phase 3 Clinical Trial Program for Advanced Dry AMD Treatment
Eye On the Cure Research NewsTrial participants will receive intravitreal injections of APL-2, or a sham procedure (placebo), monthly or every other month. The injections are made into the vitreous, the soft gel in the middle of the eye.
-
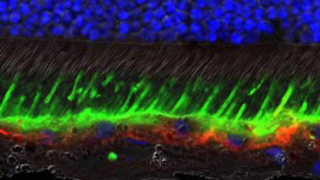
Mar 20, 2018
Best Disease Gene Therapy Advances Toward Clinical Trial
Eye On the Cure Research NewsUsing gene therapy, FFB-funded researchers at the University of Pennsylvania School of Veterinary Medicine (Penn Vet) and Perelman School of Medicine have reversed the disease process in a canine model of Best disease, an inherited form of macular degeneration that can lead to severe vision loss in humans
-
Nov 21, 2017
Stem-Cell Therapy Clinics Remain Inadequately Regulated, Pose Risk to Patients
Eye On the Cure Research NewsIf a clinic is charging for a stem-cell treatment or procedure for an IRD, it is probably not legit. The expense to the patient is a major red flag.
-
Nov 15, 2017
ProQR Doses First Participant in Its LCA10 Therapy Clinical Trial
Eye On the Cure Research NewsThe company plans to report interim, six-month study results in 2018 and 12-month results in 2019.





