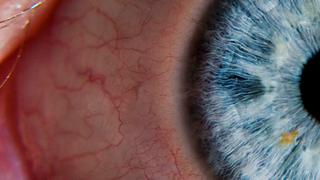
Age-Related Macular Degeneration
Age-related macular degeneration, or AMD, is the leading cause of blindness in people 55 and older in developed countries. The condition can lead to significant loss of central vision.
Jump to Section
What is Age-Related Macular Degeneration?
Age-related macular degeneration, commonly referred to as AMD, is a retinal degenerative disease that causes a progressive loss of central vision. AMD is the most common cause of blindness in individuals over the age of 55 in developed countries. More than 10 million people in the United States have AMD.
The retina is packed with photoreceptors, the cells that enable us to see. Photoreceptors convert light into electrical impulses, which are transferred to the brain via the optic nerve and converted into the images we see. The macula is a small region in the center of the retina that’s rich in cones, the photoreceptors that enable a person to perceive fine details (e.g., read, recognize faces), colors, and objects in daylight or lighted conditions. Central vision loss from AMD occurs when cone photoreceptors in the macula degenerate.
The greatest risk factors for AMD are aging and smoking. An unhealthy diet and unprotected sunlight exposure can also increase AMD risk. Genetics is also a risk factor.
Symptoms
People with AMD may first notice a blurring of central vision, especially during tasks such as reading or sewing. Also, straight lines may appear distorted or warped. As the disease progresses, blind spots may form within the central field of vision. In most cases, if one eye has AMD, the other eye has the condition or is at risk of developing it. The extent of central vision loss varies and can depend on the type of AMD — dry or wet.
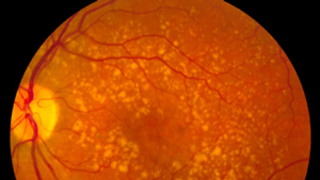
Most people with AMD start off with the dry form. In many cases, people will not experience vision loss from the condition. In some cases, dry AMD can progress and cause varying degrees of central vision loss. Advanced dry AMD is sometimes referred to as geographic atrophy.
The hallmark of dry AMD is the accumulation of tiny protein and fat deposits known as drusen underneath the retina. Many people have drusen, which do not affect vision. However, certain types of drusen may interfere with the health of the macula, causing progressive degeneration of the photoreceptor cells and vision loss.
About 10-15 percent of people with dry AMD will develop the wet form in one or both eyes. With wet AMD, abnormal blood vessels grow beneath the macula. These vessels leak blood and fluid into the macula and damage photoreceptor cells. Wet AMD often progresses rapidly and causes substantial loss of central vision, if left untreated.
Inheritance
Researchers have discovered that genetics can play a role in AMD risk. In 2005, three groups of researchers, including a team funded by the Foundation, discovered that a gene called Complement Factor H (CFH) is linked to at least 50 percent of all cases of AMD. Since that breakthrough, researchers have found several other genes linked to AMD. CFH and many of the other AMD genes are involved in the innate immune system, which fights off infection. Scientists believe that over activity of the innate immune system increases AMD risk.
While genetics can play a role in AMD risk, people with low risk genetics can still get the condition. Likewise, people with high risk genetics may not get AMD.

Make a lasting impact on retinal research. Leave a gift to the Foundation in your will or trust.
Treatment
The Age-Related Eye Disease Study (AREDS) — a landmark investigation conducted by the National Eye Institute (NEI) — determined that a specific antioxidant supplementation can slow the progression of AMD. The AREDS2 formulation is an over-the-counter antioxidant supplement recommended for people who are at risk of developing more advanced forms of dry or wet AMD.
The AREDS2 formulation contains the following nutrients:
500 milligrams (mg) of vitamin C
400 international units of vitamin E
80 mg zinc as zinc oxide
2 mg copper as cupric oxide
10 mg lutein and 2 mg zeaxanthin
Several therapies are now available for the wet form of AMD. Most involve regular ocular injections to halt the growth of leaky, vision-robbing blood vessels.
EYLEA™ (alflibercept) — Regeneron’s wet AMD treatment, Eylea, blocks the development of unhealthy blood vessels underneath the retina. Regeneron reports that in clinical trials, Eylea treated wet AMD as effectively as Lucentis, but with fewer intraocular injections. Typically, patients are treated monthly with Eylea for three months and every other month thereafter. Eylea was FDA approved in 2011.
Lucentis™ (ranibizumab) — Developed by Genentech, Lucentis is effective in reducing the risk of losing vision from the abnormal blood vessel growth under the retina associated with wet AMD. The treatment was approved by the FDA and made available in 2006. A two-year study showed that 95 percent of people with wet AMD who received monthly injections of Lucentis experienced no significant loss in visual acuity from baseline. Genentech also reported moderate visual improvement in 24.8 percent of participants treated with a 0.3 mg dose of Lucentis and 33.8 percent of participants treated with a 0.5 mg dose.
A colorectal-cancer drug called Avastin® — a drug similar to Lucentis — has been used “off-label” by some ophthalmologists to treat wet AMD. The NEI completed a large-scale, two-year clinical study comparing Avastin and Lucentis. Results of the study showed that the drugs were similar in safety and efficacy.
Next Section
Read the Most Recent Research on Age-Related Macular Degeneration
Latest News
-

Apr 16, 2024
Eye on the Cure Podcast | Episode 64: Dr. Kapil Bharti
Foundation PodcastsKapil Bharti, PhD, a senior investigator and director of the Intramural Research Program at the National Eye Institute (NEI), talks with host Ben Shaberman about his clinical trial underway for an innovative cell therapy for geographic atrophy (advanced dry age-related macular degeneration) as well as a clinical trial of the drug metformin for Stargardt disease.
-

Mar 8, 2024
Eye on the Cure Podcast | Episode 63: Dr. Hema Ramkumar
Foundation PodcastsHema Ramkumar, MD, a retinal specialist, surgeon, and founder of the company Oculogenex, talks with host Ben Shaberman about her emerging therapy that targets mitchondria to slow the progression of intermediate dry age-related macular degeneration and potentially other retinal diseases. She also discusses her experiment that will put mice in space so researchers can gain a better understanding of retinal degeneration.
-

Sep 6, 2023
Therapeutic Targets for Dry AMD: A Foundation Fighting Blindness Webinar for Eye Care Professionals
Press ReleasesThe free, online event will feature a lecture from David Boyer, MD, a Senior Partner at Retina-Vitreous Associates Medical Group and dry AMD expert.
-

Jan 6, 2023
The Retinal Degeneration Fund invests in a $78M Series B for Perceive Biotherapeutics
The Foundation in the NewsThe investment supports the clinical development of two novel programs, both with the aim of being protective against vision loss.
-

Oct 13, 2022
Economic Burden of Ageing Eye conditions estimated on the scale of up to billions in USA, Germany and Bulgaria
The Foundation in the NewsOn World Sight Day 2022, Retina International is presenting data from a report on its study into the Socio-economic Impact of late-stage age-related macular degeneration (AMD) in Bulgaria, Germany, and USA.
-

Aug 4, 2020
Foundation Insights Forum – July 30, 2020
Insights ForumThe Foundation Fighting Blindness is pleased to provide an audio recording and full transcript of the Insights Forum, our quarterly conference call providing updates to the inherited retinal disease community. The call took place on July 30, 2020.
-
Mar 31, 2020
COVID-19 Resources
Foundation NewsThe Foundation Fighting Blindness is closely monitoring the COVID-19 situation and its impact on the IRD community.
-
Feb 6, 2020
Foundation Insights Forum – January 31, 2020
Insights ForumThe Foundation Fighting Blindness is pleased to provide an audio recording and full transcripts of the Insights Forum, our quarterly conference call providing updates to the inherited retinal disease community. The call took place on January 31, 2020.
-

Feb 6, 2020
ProQR Therapeutics Teams Up with the Foundation Fighting Blindness and Blueprint Genetics to Support the My Retina Tracker® Program for People Living with Inherited Retinal Diseases
Press ReleasesMy Retina Tracker Program is the highest volume IRD genetic testing program in the U.S.
-
Nov 8, 2019
Foundation Insights Forum – October 30, 2019
Insights ForumThe Foundation Fighting Blindness is pleased to provide an audio recording and full transcript of the Insights Forum, our quarterly conference call providing updates to the inherited retinal disease community. The call took place on October 30, 2019.
-
Oct 2, 2019
Blueprint Genetics, InformedDNA and the Foundation Fighting Blindness launch an open access program for patients with inherited retinal disease in the United States
Press ReleasesThe program will offer patients with inherited retinal disease no-cost genetic testing and genetic counseling in the United States. Look for updated information on how to participate to be posted in mid-October, with program registration starting shortly thereafter.
-
Aug 16, 2019
Foundation Fighting Blindness Investing Nearly $6.5 Million in New Grants
Foundation NewsThe newly funded research efforts include several therapies that have strong potential to treat a wide range of inherited retinal diseases.
-
Jul 11, 2019
Sustained Suppression of VEGF: Looking Forward and Looking Back
Industry NewsThe results of the LADDER trial demonstrate a major step forward but introduce many questions.
-
Jul 11, 2019
Gene Therapy Trials for Wet Age-Related Macular Degeneration
Industry NewsAfter a series of failed trials, current research suggests that the next generation of gene therapies for wet age-related macular degeneration holds promise. Learn how investigators are inducing the body’s cells to administer their own therapeutic response to disease activity.
-

May 9, 2019
Foundation Fighting Blindness Endorses 'Eye Bonds' Legislation
Press ReleasesBipartisan Bill Will Stimulate Up to $1 Billion in New Funding for Blindness Research
-
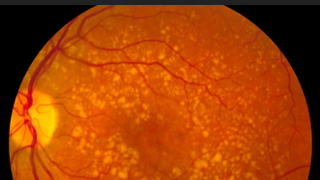
Mar 7, 2019
The Foundation Fighting Blindness and Dr. H. James & Carole Free Collaborate to Combat AMD
Foundation NewsAge-related macular degeneration (AMD) is the leading cause of blindness for people over 50 years of age in developed countries.
-

Jul 19, 2018
Foundation Fighting Blindness Urges Congress to Pass ‘Eye-Bonds’ Legislation
Press ReleasesBill Introduced in U.S. House Would Speed Up Cures for Blindness
-

Jun 8, 2018
Foundation Fighting Blindness and CheckedUp® Partner to Educate Retinal-Disease Patients About Research, Resources, and Emerging Therapies During Doctor Visits
Press ReleasesThe Foundation Fighting Blindness (the Foundation) and CheckedUp have formed a collaborative partnership to deliver patient-friendly diagnostic and disease-management information to people with retinal diseases such as age-related macular degeneration, retinitis pigmentosa, and Stargardt disease during their visits to eye doctors.
-
Apr 4, 2011
Researchers Take another Critical Step toward Using Skin Cells to Treat Retinal Disease
Foundation NewsA research team funded by the Foundation Fighting Blindness used an innovative repair technique to correct the disease-causing genetic defect in stem cells derived from a person’s skin — stem cells that hold the potential to treat their retinal degenerative condition.
Latest Research
-
Jan 8, 2024
Age-Related Macular Degeneration Research Advances
Retinal Disease Research AdvancesRecent developments in research on age-related macular degeneration.
-

Aug 22, 2023
FDA Approves 8 MG Dosing of Eylea for Wet AMD, Diabetic Macular Edema, and Diabetic Retinopathy
Eye On the Cure Research NewsNew, higher treatment dose reduces frequency of eye injections for patients
-

Aug 5, 2023
IZERVAY Approved by FDA for Treatment of Geographic Atrophy Secondary to Advanced Dry AMD
Eye On the Cure Research NewsIn two Phase 3 clinical trials, IZERVAY slowed the growth rate of lesions, the regions of cell loss in the central retina, associated with geographic atrophy (GA)
-

Jul 28, 2023
Belite Bio Doses First Patient in Phase 3 Clinical Trial for Advanced Dry AMD (GA) Therapy
Eye On the Cure Research NewsThe company’s emerging, oral treatment is designed to slow vision loss
-

Jun 12, 2023
Beacon Therapeutics to Advance XLRP, Cone-Rod Dystrophy, and Dry AMD Gene Therapies
Eye On the Cure Research NewsAn emerging XLRP gene therapy acquired from AGTC is the company’s lead clinical program
-
Apr 19, 2023
Geographic Atrophy: The Advanced Form of Dry AMD
Science EducationGeographic atrophy causes significant central vision loss and remains an unmet medical need.
-

Feb 18, 2023
FDA Approves Apellis’ SYFOVRE™ for the Treatment of Geographic Atrophy Secondary to Age-Related Macular Degeneration (AMD)
Eye On the Cure Research NewsGeographic atrophy (GA) is the advanced form of dry age-related macular degeneration (AMD) which causes blindness in one million people in the United States and 5 million worldwide.
This is the first time that a treatment has been approved for this leading cause of blindness.
-

Sep 8, 2022
Iveric Bio’s Zimura for Advanced Dry AMD Meets Endpoint in Second Phase 3 Clinical Trial
Eye On the Cure Research NewsThe company plans to seek FDA approval for the emerging therapy in first quarter of 2023
-

Jul 22, 2022
Apellis’ Treatment for Advanced Dry AMD to Receive Priority Review from FDA
Eye On the Cure Research NewsThere are currently no FDA-approved therapies for geographic atrophy (GA), the advanced form of dry AMD.
-

May 13, 2022
Enhanced Implantable Miniature Telescope Moving into Clinical Trial for AMD Patients
Eye On the Cure Research NewsThe device is designed for people with late-stage AMD who have no treatment options
-

Feb 1, 2022
Genentech-Roche Receives FDA Approval for Vabysmo for Treatment of Wet AMD and DME
Eye On the Cure Research NewsThis new treatment can reduce the frequency of injections into the eye
-
.png)
Jan 28, 2022
Kriya Therapeutics Licenses Emerging Foundation-Funded Dry AMD Gene Therapy
Eye On the Cure Research NewsThe Foundation Fighting Blindness is funding Bärb Rohrer, PhD, Medical University of South Carolina, to evaluate safety and efficacy of the gene therapy in a large animal model
-

Sep 10, 2021
Apellis to Seek FDA Approval of its Dry AMD Drug
Eye On the Cure Research NewsKnown as APL-2, the drug met its primary endpoint in the OAKS clinical trial but not the DERBY trial
-

Sep 9, 2021
Optogenetics: Hope for Vision Restoration for Advanced Retinal Diseases
Eye On the Cure Research NewsEarly, encouraging results from two human studies — trials launched by Bionic Sight and GenSight — are putting optogenetic therapies in the spotlight for patients with advanced vision loss from retinal conditions.
-
Apr 9, 2021
Foundation Invests $5.5 Million in Seven New Translational Research Projects
Eye On the Cure Research NewsProjects target a variety of conditions including: age-related macular degeneration, Stargardt disease, retinitis pigmentosa, and Usher syndrome type 3A
-

Jan 26, 2021
Genentech’s Faricimab Performs Well in Phase 3 Clinical Trials for Wet AMD
Eye On the Cure Research NewsCompany will seek marketing approval for the emerging therapy
-

Jan 7, 2021
REGENXBIO’s Wet AMD Gene Therapy Moving into Phase 3 Trials
Eye On the Cure Research NewsIn a Phase ½ clinical trial, the emerging treatment reduced the burden of regular anti-VEGF injections
-

May 28, 2020
Genentech’s Port Delivery System for Wet AMD Meets Primary Endpoint in Phase 3 Clinical Trial
Eye On the Cure Research NewsTiny implant provides continual delivery of anti-VEGF treatment for six months
-

May 15, 2020
Positive Interim Clinical Trial Results for Adverum’s Wet AMD Gene Therapy
Eye On the Cure Research NewsThe gene therapy is designed to reduce the treatment burden for patients
-

Apr 15, 2020
Eye Disease Studies Suggest Mediterranean Diet is Good for the Brain
Eye On the Cure Research NewsA healthy diet is also important for people with retinal conditions
-

Feb 7, 2020
Genetic Testing for Inherited Retinal Diseases through the Foundation’s Open Access Program
Science EducationThe benefits of genetic testing for IRD patients, how to participate in the Foundation’s Open Access program, and what to expect from the genetic testing process.
-

Dec 17, 2019
First Human Study in US for Induced Pluripotent Stem Cells to be Launched for Advanced Dry AMD
Eye On the Cure Research NewsCurrently there are no treatments for Advanced Dry AMD, also known as GA, which can lead to significant central vision loss
-

Nov 7, 2019
AGTC Announces Development of Stargardt Disease Gene Therapy
Eye On the Cure Research NewsDual-vector delivery system designed to deliver the large ABCA4 gene
-

Oct 29, 2019
Iveric Bio’s Therapy Slows Retinal Degeneration in Phase 2b Trial for Dry AMD
Eye On the Cure Research NewsZimura inhibits a complement protein known as C5, a component of the immune system that, when overactive, can cause retinal degeneration.
-

Oct 10, 2019
FDA Approves New Wet AMD Treatment that can be Administered Every Three Months
Eye On the Cure Research NewsBeovu from Novartis reduces treatment burden for wet AMD patients
-
Jun 17, 2019
The Retina is a Proving Ground for a Broad Range of Neurological Therapies
Science EducationRetinal research paves the way for new treatments for the entire neurological system.
-

May 20, 2019
Dr. Don Zack Honored for Research Contributions by ARVO and the Foundation Fighting Blindness
Eye On the Cure Research NewsDr. Zack is a member of the Foundation’s Scientific Advisory Board and chairs its Cellular Molecular Mechanisms of Disease study section.
-

May 9, 2019
Eye Bonds Re-Introduced to New Congress: Potentially $1 Billion in Government-Backed Funding for Eye Research
Eye On the Cure Research NewsEye Bonds provide the opportunity to advance, and accelerate development for, more promising treatments into and through clinical trials and out to the people who need them.
-

May 3, 2019
ARVO 2019: Robot-Assisted Surgery in Clinical Trial for Retinal Disease
Eye On the Cure Research NewsA video from a clinical trial of robot-assisted surgery demonstrates the potential benefits of robot-assisted subretinal injections.
-

Apr 30, 2019
REGENXBIO’s Gene Therapy for Wet AMD Performing Encouragingly in Human Study
Eye On the Cure Research NewsUnlike current treatments requiring multiple injections, REGENXBIO’s gene therapy is administered as a one-time subretinal injection to the affected eye.
-

Mar 23, 2019
First Gene Therapy for Dry AMD Moves into Clinical Trial in UK
Eye On the Cure Research NewsGyroscope Therapeutics, a gene-therapy development company in the UK, has launched the first gene therapy clinical trial for the dry form of age-related macular degeneration (AMD).
-

Jan 29, 2019
The Foundation Receives a $100,000 Research Grant from Sofia Sees Hope
Eye On the Cure Research NewsSofia Sees Hope, a nonprofit dedicated to finding treatments and cures for people with Leber congenital amaurosis (LCA) and other inherited retinal diseases (IRDs), has made a $100,000 donation to the Foundation Fighting Blindness to support therapy development and genetic testing.
-
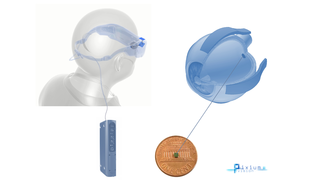
Jan 17, 2019
Pixium's PRIMA Bionic Vision System Restores Central Vision in Dry AMD Clinical Trial
Eye On the Cure Research NewsThe French bioelectronics company Pixium Vision has reported that its PRIMA bionic vision system has restored some central vision in patients with advanced dry age-related macular degeneration (AMD) participating in a clinical feasibility trial.
-

Nov 21, 2018
Acucela Enrolling Patients in Phase 3 Trial for Stargardt Disease Treatment
Eye On the Cure Research NewsThe Seattle biotech Acucela is now enrolling participants in its Phase 3 clinical trial for emixustat hydrochloride, an emerging oral drug for slowing vision loss in people with Stargardt disease, an inherited form of a macular degeneration.
-

Nov 2, 2018
Foundation Invests $2.5 Million in Search for Elusive Retinal Disease Genes and Mutations
Eye On the Cure Research NewsSince 1989 genetic researchers, many funded by the Foundation, have identified approximately 270 genes linked to IRDs. In most cases, defects in a single gene can cause a retinal disease and vision loss.
-

Sep 18, 2018
Apellis Launches Phase 3 Clinical Trial Program for Advanced Dry AMD Treatment
Eye On the Cure Research NewsTrial participants will receive intravitreal injections of APL-2, or a sham procedure (placebo), monthly or every other month. The injections are made into the vitreous, the soft gel in the middle of the eye.
-

Sep 11, 2018
FFB Congratulates RPE65 Gene Therapy Researchers for Champalimaud Award
Eye On the Cure Research NewsOn September 4, 2018, seven researchers, including six previously funded by the Foundation, were recognized with the prestigious 2018 Antonio Champalimaud Vision Award for their contributions to the advancement of blindness-reversing RPE65 gene therapies.
-

Aug 22, 2018
Ophthotech is Advancing an Impressive Portfolio of Cutting-Edge Therapies for Retinal Diseases
Eye On the Cure Research NewsThe company is taking on a multi-track strategy that includes retinal gene-therapy development, including delivery of over-sized genes and design of a two-step process of gene knockdown and replacement for autosomal dominant conditions.
-
Aug 15, 2018
FFB Provides Four Career Development Awards to Up-and-Coming Clinical Researchers
Eye On the Cure Research NewsEach recipient will receive a total of $375,000 over five years to help build an independent research program in addition to their clinical practices.
-
Aug 6, 2018
FFB Funding More than $2 Million in New Research
Eye On the Cure Research NewsSeventy scientists submitted requests for funding.
-

Jul 20, 2018
Call to Action: Ask Congress to Support $1 Billion in Eye Research
Eye On the Cure Research NewsCall to Action: Ask Congress to Support $1 Billion in Eye Research
-
Jul 5, 2018
Retinal Regeneration: Releasing Your Inner Salamander
Eye On the Cure Research NewsMany research groups from around the world are investigating ways to create new photoreceptors from stem cells for transplantation into the retina for vision restoration.
-
Jun 22, 2018
VISIONS2018 Live Stream
Eye On the Cure Research NewsWatch recorded sessions from VISIONS2018.
-
May 4, 2018
ARVO 2018: Port Delivery System Designed to Reduce Burden of Lucentis Injections for Wet AMD
Eye On the Cure Research NewsDr. Christopher Brittain, Genentech medical director, discusses his company’s port delivery system, a tiny capsule implanted into the eye, for delivery of Lucentis® over a period of a few months.
-

May 3, 2018
ARVO 2018: Dr. Stephen Daiger Reports on the State of Genetic Testing for Inherited Retinal Diseases
Eye On the Cure Research NewsARVO 2018: Dr. Stephen Daiger Reports on the State of Genetic Testing for Inherited Retinal Diseases
-

May 2, 2018
ARVO 2018: Dr. Steve Rose Reports on CRISPR/Cas9 for Inherited Retinal Diseases
Eye On the Cure Research NewsFFB’s own Dr. Steve Rose, chief scientific officer, reviews our commitment to funding and exploring CRISPR/Cas9 gene editing for inherited retinal disease.
-

May 1, 2018
ARVO 2018: Studies Show Mediterranean Diet May Lower Advanced AMD Risk
Eye On the Cure Research NewsSeveral research studies have suggested that the Mediterranean diet (MeDi) - which is rich in fruits, vegetables, legumes, cereals, and fish - may be good for your health, including lowering your risk of an early visit from the grim reaper.
-

Apr 25, 2018
ARVO 2018: World's Largest Show and Tell for Innovations in Eye Research
Eye On the Cure Research NewsMore than 11,000 eye researchers from around the world — including five intrepid members from FFB’s science team — will gather to participate in what is essentially a massive “show and tell” of the latest scientific advancements.
-
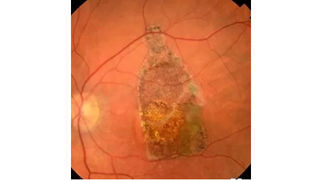
Apr 16, 2018
Retinal Patch Performs Promisingly in Clinical Trial for Dry AMD Patients
Eye On the Cure Research NewsRegenerative Patch Technologies, a company developing stem-cell-derived treatments for people with retinal diseases, has reported encouraging results for the first five patients with advanced, dry age-related macular degeneration (AMD) participating in a Phase I/IIa clinical trial for its therapy – a patch comprised of a layer of retinal pigment epithelial (RPE) cells on a synthetic scaffold.
-
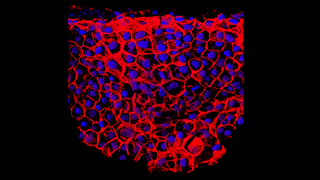
Mar 27, 2018
Vision Improvements Reported in Early Stem Cell Trial for Wet AMD
Eye On the Cure Research NewsTwo patients with advanced wet age-related macular degeneration (AMD) in a Phase I clinical trial demonstrated improved visual acuity sustained for one year after a sheet of retinal pigment epithelial (RPE) cells derived from embryonic stem cells was transplanted under their retinas. Each patient had one eye treated. Vision improvement for one patient was 29 letters or about 6 lines on an eye chart. The other had a gain of 21 letters or about 4 lines.
-
Jan 17, 2018
Clinical Trial to Launch for System Combining Optogenetics and Eyewear
Eye On the Cure Research NewsThe French biotech GenSight Biologics has received regulatory authorization in the UK to launch the PIONEER Phase 1 \ 2 clinical trial for its GS030 system — a light-sensing gene therapy (optogenetics) coupled with eyewear, which enhances visual stimulation.
-

Jan 9, 2018
Top Retinal Research Advances for 2017
Eye On the Cure Research NewsAn exciting year in fighting blindness.
-
Dec 20, 2017
History Is Made: FDA Approves Spark's Vision-Restoring Gene Therapy
Eye On the Cure Research NewsKnown as LUXTURNA™ (voretigene neparvovec), the gene therapy restored vision in a clinical trial for people between the ages of 4 and 44 with Leber congenital amaurosis (LCA) caused by mutations in the gene RPE65.
-
Nov 21, 2017
Stem-Cell Therapy Clinics Remain Inadequately Regulated, Pose Risk to Patients
Eye On the Cure Research NewsIf a clinic is charging for a stem-cell treatment or procedure for an IRD, it is probably not legit. The expense to the patient is a major red flag.
-

Oct 13, 2017
FDA Committee Unanimously Recommends Approval for Spark's RPE65 Gene Therapy - Final Decision Due in January 2018
Eye On the Cure Research NewsAn advisory committee comprised of FDA-selected experts voted unanimously – 16 to 0 – to recommend approval.
-

Sep 27, 2017
The Foundation's Investments Are Filling the Pipeline for Vision-Saving Therapies
Eye On the Cure Research NewsIn addition to funding promising biotech start-ups, the Foundation Fighting Blindness has played a critical role in developing research talent.
-
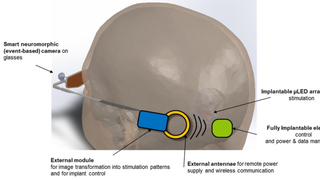
Jul 26, 2017
Scientists Receive $25 Million to Develop a Vision-Restoring System that Connects to the Brain
Eye On the Cure Research NewsThe high-tech, vision-restoring system interfaces with the visual cortex, the back of the brain where visual input is processed to create the images we see.
-
Jul 25, 2017
Foundation Fighting Blindness and 4D Molecular Therapeutics Partner to Boost Retinal Gene Therapy Development
Eye On the Cure Research NewsThe partnership will help companies and researchers quickly obtain and implement high-quality vectors for their retinal gene-therapy development efforts.
-
Jul 24, 2017
FFB-Funded Scientists Report on Nine Promising Translational Research Efforts
Eye On the Cure Research NewsThe Foundation Fighting Blindness has taken the translational challenge head on by investing more than $75 million in therapy-development projects with strong clinical-trial potential through its Translational Research Acceleration Program (TRAP), which includes Gund-Harrington Scholar Awards.
-

May 23, 2017
Forty-Four High-Impact Retinal-Research Efforts Highlighted at FFB-Casey Innovation Summit
Eye On the Cure Research NewsIn its fourth year, the meeting is becoming the world’s most comprehensive overview of the promising research underway for emerging IRD treatments.
-

May 8, 2017
FFB Funding Helps Retinal Genetics Lab Secure $2 Million Investment
Eye On the Cure Research NewsHow the Foundation Fighting Blindness (FFB) provided timely funding of $155,000 to help a lab at the University of California, San Diego (UCSD), leverage a $2 million retinal-gene discovery project.
-

Mar 22, 2017
Dr. Eliot Berson, Pioneer in Vitamin A Therapy for Retinitis Pigmentosa, Passes Away
Eye On the Cure Research NewsDr. Berson dedicated himself to clinical care and vision-saving research for people with inherited retinal diseases for five decades.
-
Mar 16, 2017
Unregulated Stem-Cell Therapy Causes Severe Vision Loss for Three Florida Women
Eye On the Cure Research News“…participation in a study for an emerging therapy that is not regulated by the FDA or another well-recognized regulatory agency like the European Medicines Agency in Europe, is fraught with dangers and can lead to unexpected serious consequences.”
-
Feb 17, 2017
AGTC Leverages Funding from the Foundation to Move Promising Treatments into Clinical Trials
Eye On the Cure Research NewsCompany Builds on FFB’s Initial Investment to Garner $265 Million in Therapy Development Funding
-
Dec 21, 2016
FFB-CRI Leads Effort to Identify Outcome Measures for Therapies in Clinical Trials
Eye On the Cure Research NewsImproved outcome measures will make clinical trials for degenerative retinal diseases — including age-related macular degeneration (AMD), the world’s leading cause of blindness in seniors, and inherited retinal conditions such as RP and Stargardt disease — less expensive to conduct and able to deliver more precise results.
-

Oct 18, 2016
Building a Wiring Diagram for the Retina to Help Researchers Save and Restore Vision
Eye On the Cure Research NewsUnderstanding the pathways of the retinal neural network — and how they are rewired with aging and disease — is helpful in trying to save and restore vision.
-

Oct 11, 2016
Nobel-Prize-Winning Stem-Cell Researcher Delivers Keynote at FFB-Funded Conference in Kyoto
Eye On the Cure Research NewsDr. Shinya Yamanka discussed his early clinical trial for iPSC-derived retinal pigment epithelial (RPE) cells for a 78-year-old woman with advanced wet age-related macular degeneration (AMD).
-
.png)
Oct 6, 2016
Embrace Your Exceptions: A Mantra for Understanding Retinal-Disease Inheritance
Eye On the Cure Research NewsThe complex and elusive nature of these conditions can also extend to the way they are passed down in families, making diagnosis and prognosis quite challenging.
-

Aug 18, 2016
Optogenetic Therapy Takes First Step Forward in Clinical Trial
Eye On the Cure Research NewsRetroSense’s optogenetic therapy is designed to restore vision to people who are completely blind from retinal degenerative diseases such as retinitis pigmentosa by bestowing light sensitivity to retinal ganglion cells, which survive after photoreceptors, the cells that make vision possible, are lost.
-

Aug 2, 2016
Pixium Vision Reports Progress in Development of Two Advanced Bionic Retina Systems
Eye On the Cure Research NewsBoth approaches show strong, near-term potential for providing meaningful vision to people who are otherwise blind from retinal diseases such as retinitis pigmentosa and age-related macular degeneration (AMD).
-
Jul 1, 2016
VISIONS 2016 — Dr. Shomi Bhattacharya Wins FFB Award for Gaining an Understanding of Variations in Vision Loss
Eye On the Cure Research NewsAt VISIONS 2016, FFB’s national conference, the Foundation honored him with its Ed Gollob Board of Directors Award for breakthrough research conducted within the past year.
-

Jul 1, 2016
VISIONS 2016 - Dr. Richard Weleber Receives FFB's Highest Research Honor, Recognized in Touching Video
Eye On the Cure Research NewsDr. Weleber became the 10th recipient of the Foundation’s highest honor, named after FFB co-founder Lulie Gund, during the opening lunch of the VISIONS 2016 conference.
-
Jun 24, 2016
A Steady Hand in Saving Vision
Eye On the Cure Research NewsSubretinal injection is the most common form of delivery for gene therapies currently in clinical trials.
-
Nov 11, 2015
Past Sun Exposure Increases AMD Risk
Eye On the Cure Research NewsResearchers from the University of Cologne in Germany and Radboud University in the Netherlands found that increased sunlight exposure during working life significantly increases risk of AMD later in life.
-

Oct 8, 2015
A Leap Forward: Spark Therapeutics Seeks FDA Approval for its Vision-Restoring Gene Therapy
Eye On the Cure Research News -

Jul 28, 2015
First AMD Patient Receives Argus II Bionic Retina
Eye On the Cure Research NewsA pioneering, 80-year-old named Ray Flynn is the first AMD patient to receive the Argus II, and he is already reporting that he can see things better in his garden.
-
Jun 27, 2015
VISIONS 2015 — Dr. José Sahel Receives Foundation's Most Prestigious Research Honor
Eye On the Cure Research NewsFor those of us supporting the drive for vision-saving treatments and cures, he’s exactly the type of person we want on our team.
-

Jun 26, 2015
VISIONS 2015 — Dr. Shannon Boye Receives FFB Award for Excellence in Gene-Therapy Research
Eye On the Cure Research NewsDr. Boye received the Foundation’s Board of Director’s Award, which was presented at VISIONS 2015, FFB’s annual conference, for achievements in retinal research.
-
May 19, 2015
ARVO 2015 Highlight: The National Eye Institute Invests $4 Million in Audacious-Goals Research
Eye On the Cure Research NewsThe mission of the program—to regenerate the neurons and neural connections in the eye and visual system—is synonymous with the Foundation’s mission to eradicate retinal diseases.
-
May 12, 2015
ARVO 2015 Highlight: A Cut-and-Paste Approach to Fixing Retinal-Disease Genes
Eye On the Cure Research NewsOne of the hot topics at ARVO this year is a rapidly advancing gene-therapy approach called clustered regularly interspaced short palindromic repeats, or CRISPR.
-

Aug 1, 2014
How Evolution is Leading to Gene Therapies for More Retinal Diseases
Eye On the Cure Research NewsAn innovative genetic-engineering approach called “directed evolution” to find optimal gene-delivery systems based on adeno-associated viruses (AAVs).
-
Jun 21, 2014
VISIONS 2014 — My Retina Tracker: Track Your Vision and Drive the Research
Eye On the Cure Research NewsThe powerful and secure system enables patients to keep track of their clinical care and vision changes. At the same time, it enables scientists to search the “de-identified” (i.e., anonymous) patient information to study conditions and identify targets for treatments, preventions and cures.
-
Jun 21, 2014
VISIONS 2014 — The Multi-Talented Dr. Shannon Boye
Eye On the Cure Research NewsDr. Boye and her research team received a $900,000 grant for a gene therapy project targeting Leber congenital amaurosis.
-
May 8, 2014
ARVO 2014: European Collaboration Developing Cross-Cutting, Vision-Saving Therapies
Eye On the Cure Research NewsSimply put, they’re creating therapies that can save vision in as many people as possible, independent of the genetic cause of disease.
-
Apr 8, 2014
Total Blindness and Non-24 Sleep Disorder
Eye On the Cure Research NewsNon-24 is a very rare condition affecting many (but not all) people who are totally blind and have absolutely no light perception. Their circadian clocks become out of sync as a result.
-
Dec 31, 2013
Nouvelle Lumière: French Bionic Retina in a Human Study
Eye On the Cure Research NewsThe French retinal implant developer Pixium quietly launched a clinical trial for its Intelligent Retinal Implant System 1 (IRIS1) in France, Austria and Germany.
-
Jul 26, 2013
Researchers Move Closer to Getting a Complete Genetic Picture of the Retina
Eye On the Cure Research NewsIdentifying the genes and proteins that play a major role in retinal health and vision is an important step in finding preventions and cures for degenerative diseases.
-
Jun 12, 2013
Patient Registries Help Advance Research for Rare Diseases
Eye On the Cure Research NewsMany registries enable patients to collect and track information about their health, so they can take an active role in managing their care.
-
May 10, 2013
Grow Your Own: Harnessing Muller Glia for Retinal Regeneration
Eye On the Cure Research NewsThere’s hope for retinal regeneration for humans, thanks to Foundation-funded researcher Dr. Thomas Reh, who is investigating how to derive new photoreceptors from retinal cells called Muller glia.
-
May 7, 2013
Retinal Regeneration is Major Focus of NEI's Audacious Goal
Eye On the Cure Research NewsThe goal, “to regenerate the neurons and neural connections in the eye and visual system,” is exactly what people with retinal diseases need to save and restore their vision.
-

Apr 30, 2013
Researcher Revolutionized Fight Against Blindness and Cancer
Eye On the Cure Research NewsA profile on Dr. Robert Langer, a medical researcher who has received dozens of awards, accolades and honorary degrees, including, recently, FFB’s Visionary Award.
-
Mar 8, 2013
Staying Alive: Saving Retinal Cells to Preserve Vision
Science EducationSometimes, saving vision simply comes down to keeping retinal cells alive, or at least slowing their degeneration.
-

Feb 18, 2013
History in the Making
Eye On the Cure Research NewsMore good news about treatments and technological advances for restoring vision for people with retinal diseases.
-

Jan 15, 2013
Why Face Recognition Can Be Difficult with Central Vision Loss
Science EducationDiseases such as age-related macular degeneration, cone-rod dystrophy and Stargardt disease cause scotomas, or blind spots, which often have devastating effects on central vision. They cause gaps in a person’s visual field, making it difficult to see words in a book, images on a computer monitor or TV and the features of someone’s face.
-
Jul 27, 2012
Tempering the Immune System to Save Vision
Science EducationGreat progress is being made in identifying targets for therapies, including using the body’s own protective systems to keep the photoreceptors alive.
-

Jul 17, 2012
Tiny Implantable Telescope Can Restore Some Central Vision in AMD
Eye On the Cure Research NewsKnown as the implantable miniature telescope (IMT), the device is surgically placed in the eye, where it focuses images onto the area of the patient’s retina that is still functional.
-
Jun 19, 2012
Have I Got a Cure for You! Debunking an Alleged Treatment on the Internet
Eye On the Cure Research NewsHow do you know if a treatment is legit? There should be preclinical and clinical trial data published in a peer-reviewed journal on research for the treatment.
Related Resources
-

May 2, 2023
Mental Health Awareness: Shattering the Stigma
Beacon StoriesAthletes around the world navigate the challenges of daily life and rigorous training, but what if a factor came into play that you had less control over? What if, one day, something changed in your vision?
-

Apr 10, 2023
Racing for AMD
Beacon StoriesSean was diagnosed with dry age-related macular degeneration (AMD) in his 40s. Now Sean is using his AMD as an opportunity to educate others about the spectrum of blinding diseases while fundraising for the Foundation Fighting Blindness.
-

Nov 22, 2021
Racing for a Reason
DIY Campaign Success StoriesSean Teare has completed a marathon and a triathlon as DIY fundraisers for the Foundation Fighting Blindness.
-

Dec 3, 2020
Janssen Acquires Rights to Novel Gene Therapy, Pioneering Treatment Solutions for Late-Stage Age-Related Macular Degeneration
Partner NewsAcquisition significantly expands Janssen’s eye disease portfolio and strengthens its gene therapy capabilities. Late-stage AMD affects millions of people with no effective treatments currently available .
-

Feb 5, 2020
Al Linde “Keeps on Rolling” with Macular Degeneration
Beacon StoriesAl Linde has learned to “just roll with it” throughout his life to overcome many challenges.




