-
Eye on the Cure Podcast | Episode 64: Dr. Kapil Bharti
Foundation Podcasts -
Phase 3 Clinical Trial of NAC Launched for RP Patients
Eye On the Cure Research News -
ViGeneron Launches Clinical Trial of Gene Therapy for RP Caused by CNGA1 Mutations
Eye On the Cure Research News
-
Ocugen to Launch Phase 3 Clinical Trial of Modifier Gene Therapy for RP
Eye On the Cure Research News -
What Does “Blindness” Really Mean?
Beacon Stories
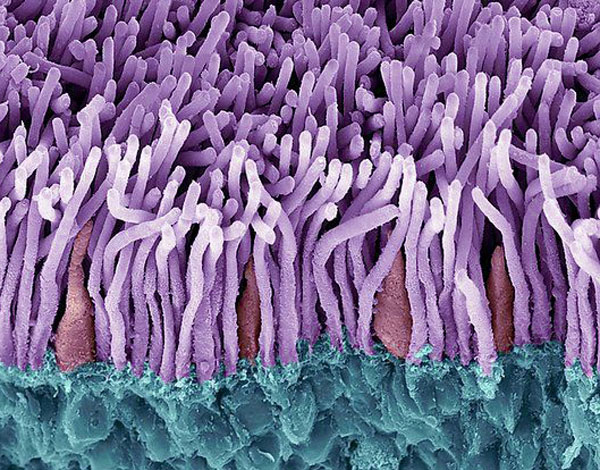
Our Impact
The Foundation is committed to finding treatments and cures for people affected by blinding retinal diseases.
-
1st gene therapy
for any inherited disease to be approved by the FDA
learn more -
$915M+ dollars raised
since 1971
learn more -
35 VisionWalk events
throughout the United States
learn more -
93 grants awarded
in 2023 – over 96 investigators, 71 institutions
Learn more
The Foundation in the News
-
Apr 11, 2024
ViGeneron Launches Clinical Trial of Gene Therapy for RP Caused by CNGA1 Mutations
Eye On the Cure Research News -
Apr 8, 2024
Ocugen to Launch Phase 3 Clinical Trial of Modifier Gene Therapy for RP
Eye On the Cure Research News -
Apr 8, 2024
What Does “Blindness” Really Mean?
Beacon Stories

Living with vision loss can be difficult, but you don't have to do it alone
View local chaptersGet Involved
-
Take important steps toward a cure by participating in a 5k VisionWalk
Join a Walk -
Join people coming together to find a cure from all over the country
Search Our Events -
Make a difference today and support promising research by donating a one-time gift
Donate Now




.png)






